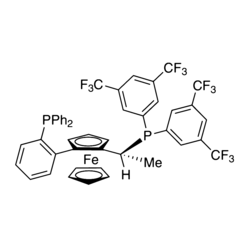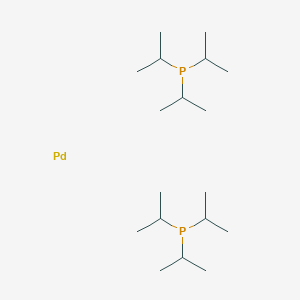
Bis[dicyclohexyl(2;,4;,6;-triisopropyl-[1,1':3',1"-terphenyl]-2--yl) phosphane]palladium(II) dichloride, 95% | Pd(Cy*Phine)2Cl2

Modern Approaches in the Synthesis of Noble Metal Porphyrins for Their Practical Application (Review) | SpringerLink

Palladium-Catalyzed Coupling of Hydroxylamines with Aryl Bromides, Chlorides, and Iodides | Organic Letters

Hyper-stable organo-EuIII luminophore under high temperature for photo-industrial application | Scientific Reports

Phosphine Oxides from a Medicinal Chemist's Perspective: Physicochemical and in Vitro Parameters Relevant for Drug Discovery | Journal of Medicinal Chemistry
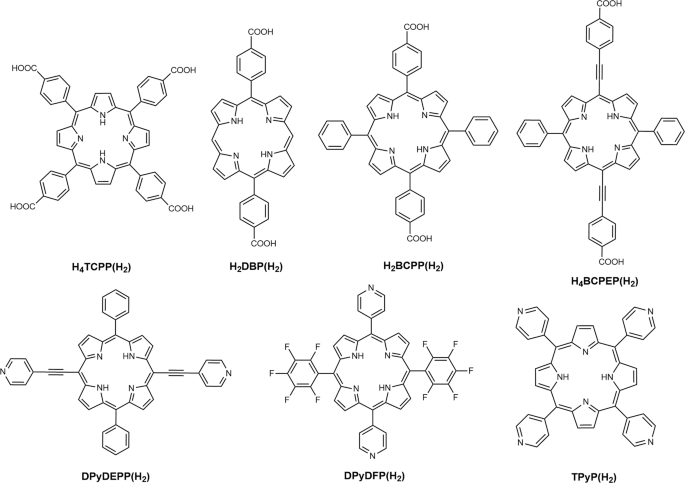
Physical properties of porphyrin-based crystalline metal‒organic frameworks | Communications Chemistry
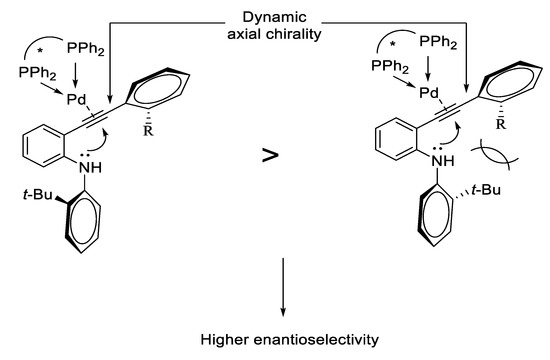
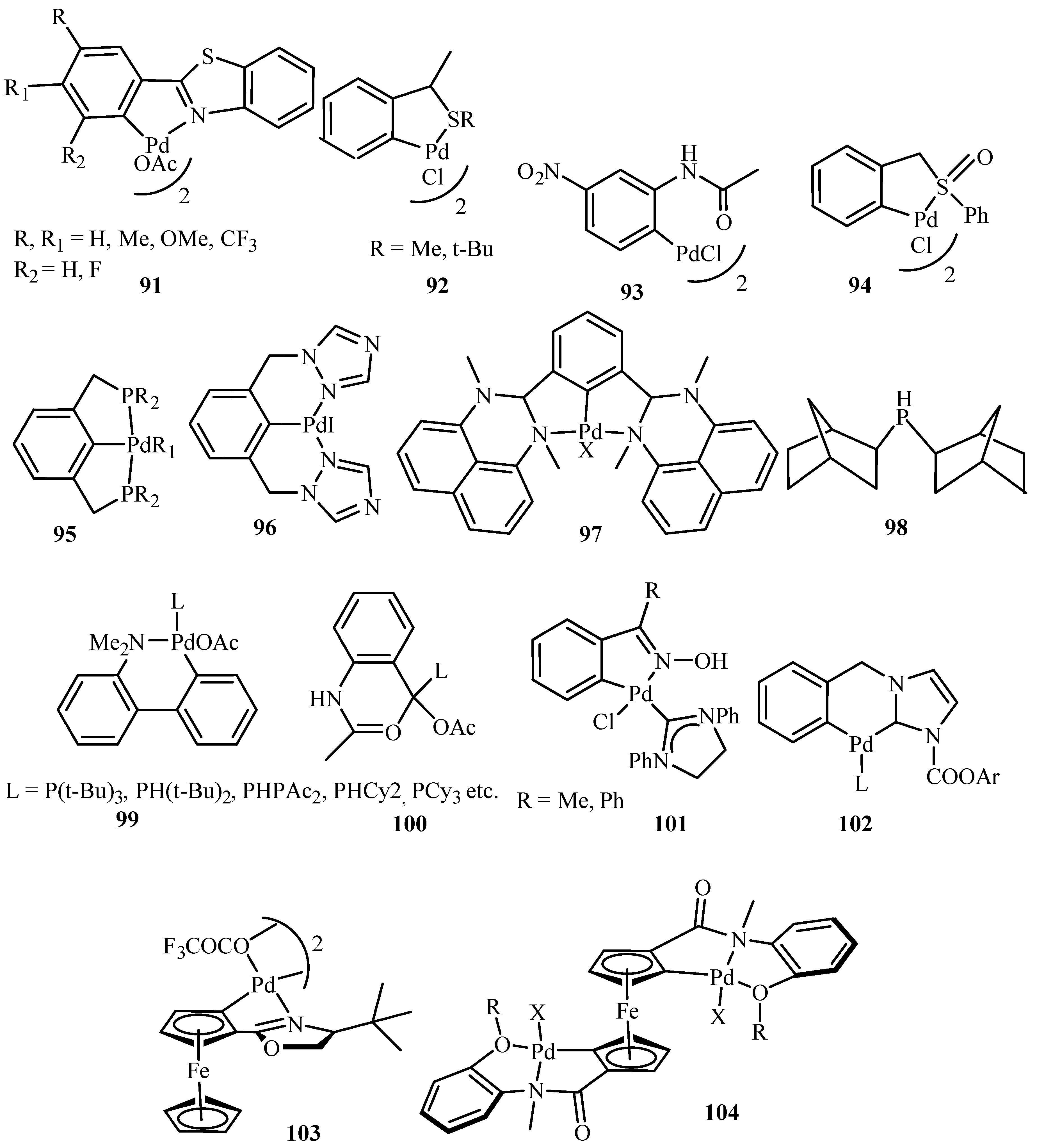
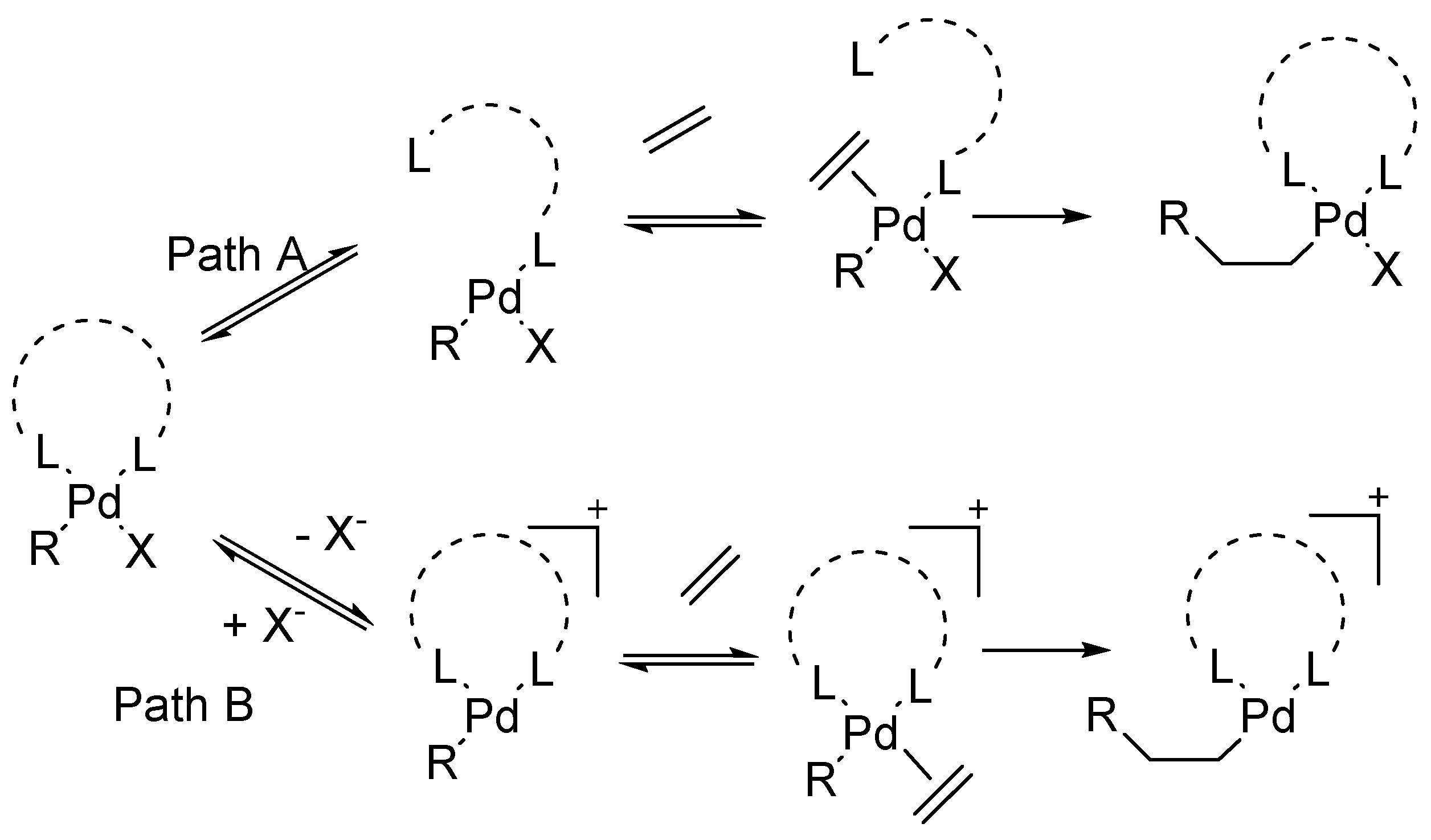
Catalysts | Free Full-Text | Synthesis of Indoles via Intermolecular and Intramolecular Cyclization by Using Palladium-Based Catalysts | HTML

NiXantphos: A Deprotonatable Ligand for Room-Temperature Palladium-Catalyzed Cross-Couplings of Aryl Chlorides | Journal of the American Chemical Society

ABC‐Type meso‐Triaryl‐Substituted Subporphyrins - Yoshida - 2014 - European Journal of Organic Chemistry - Wiley Online Library
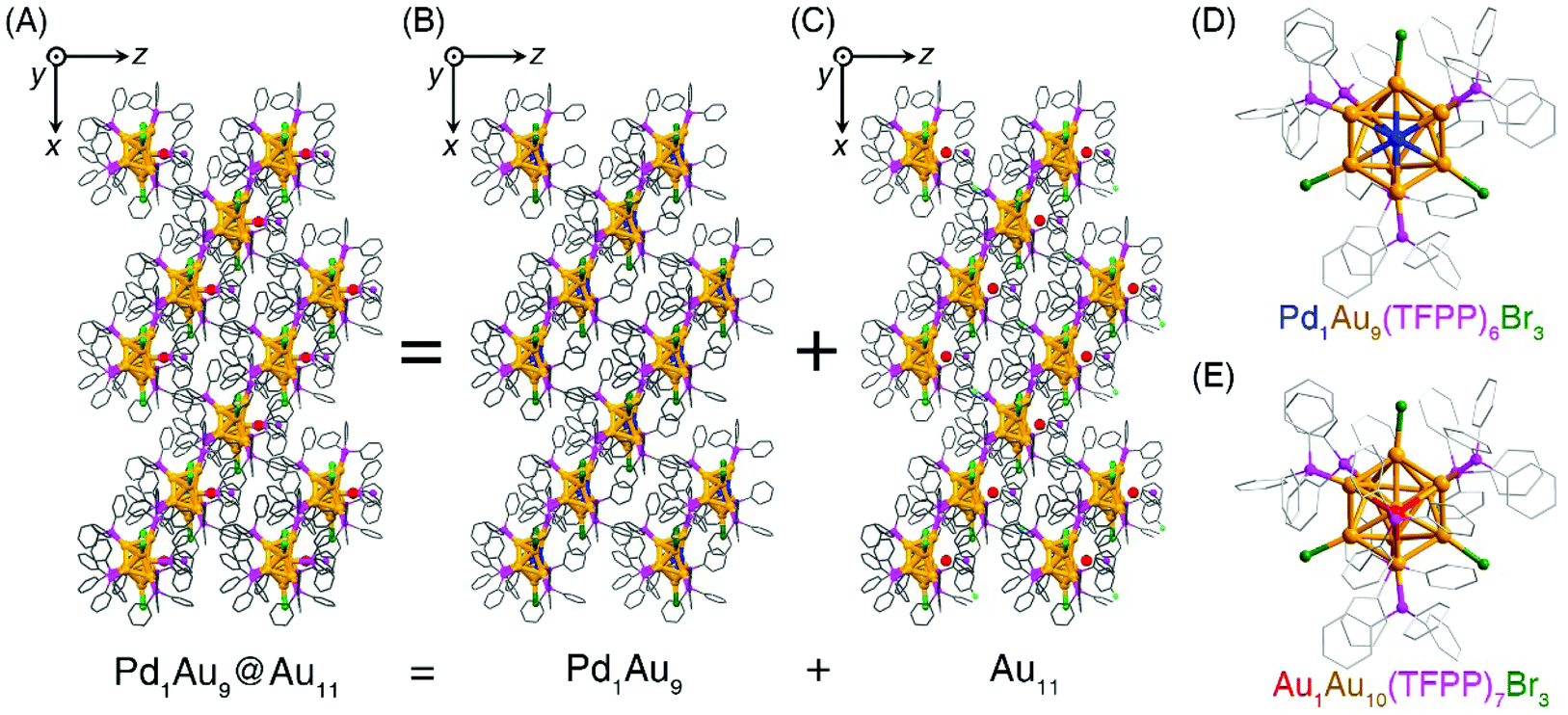
Cocrystallization-driven stabilization of metastable nanoclusters: a case study of Pd 1 Au 9 - Nanoscale (RSC Publishing) DOI:10.1039/D1NR00721A
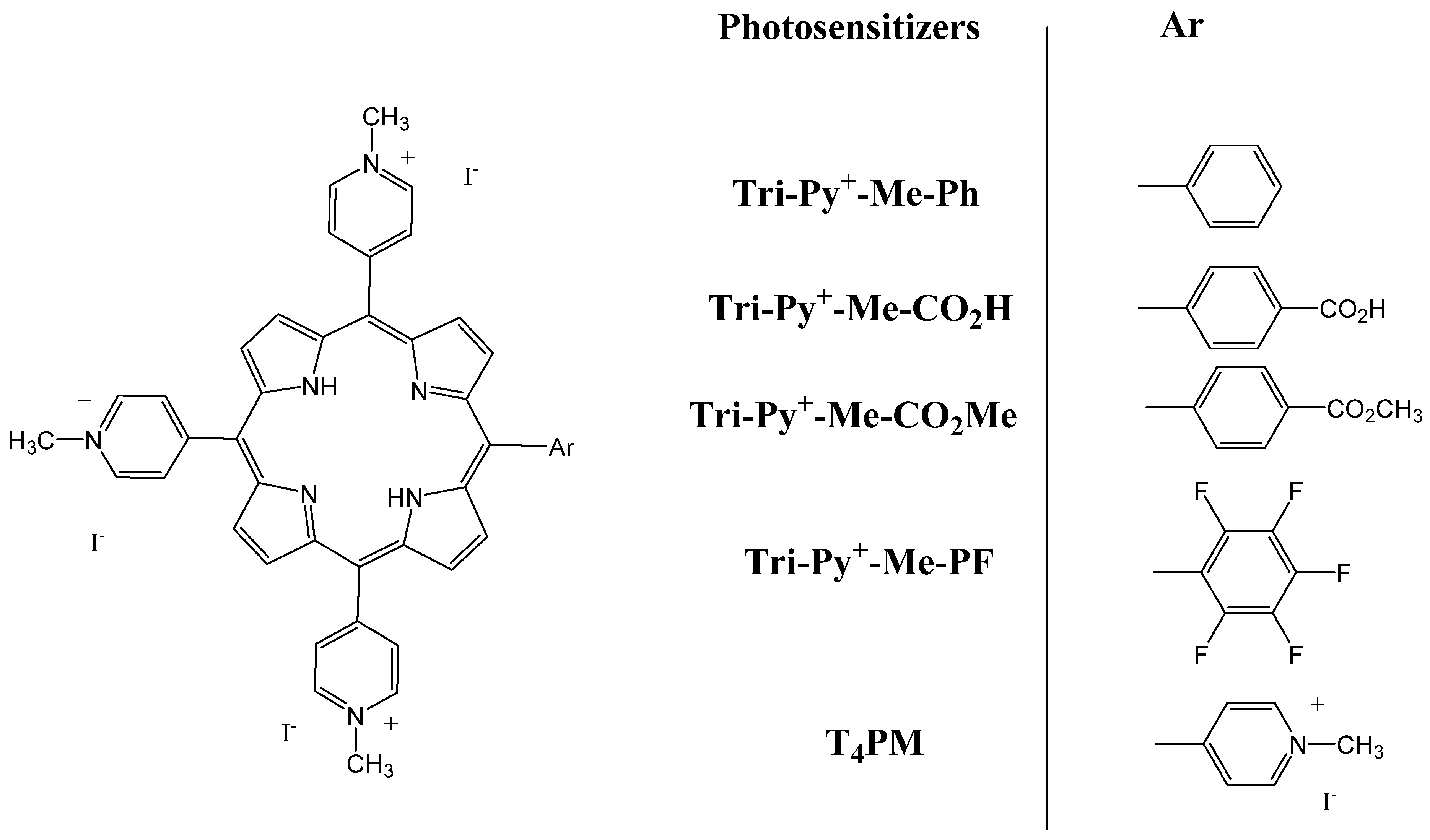
Molecules | Free Full-Text | Porphyrin/Chlorin Derivatives as Promising Molecules for Therapy of Colorectal Cancer | HTML

Palladium-Catalyzed C(sp3)–H Arylation of Diarylmethanes at Room Temperature: Synthesis of Triarylmethanes via Deprotonative-Cross-Coupling Processes | Journal of the American Chemical Society

Metal–Organic Frameworks with Low‐Valent Metal Nodes - Sikma - Angewandte Chemie - Wiley Online Library
![14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/14221-01-3.png)
14221-01-3・Tetrakis(triphenylphosphine)palladium(0)・206-18391・202-18393・204-18392[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

Interactions of porphyrins with DNA: A review focusing recent advances in chemical modifications on porphyrins as artificial nucleases - ScienceDirect

Recent Developments in the Preparation and Chemistry of Metallacycles and Metallacages via Coordination | Chemical Reviews