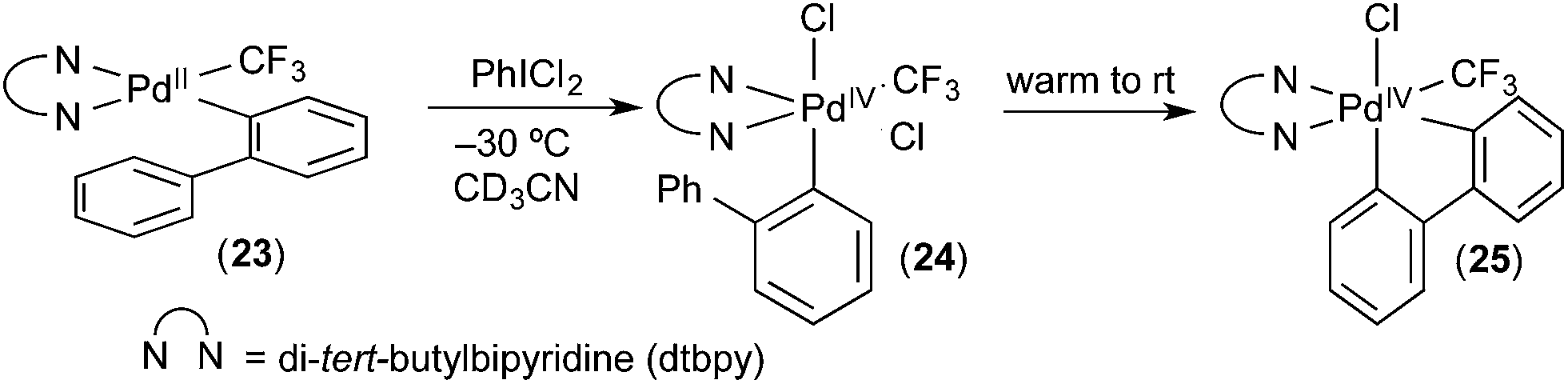
Molecules | Free Full-Text | Hypervalent Iodine Reagents in High Valent Transition Metal Chemistry | HTML
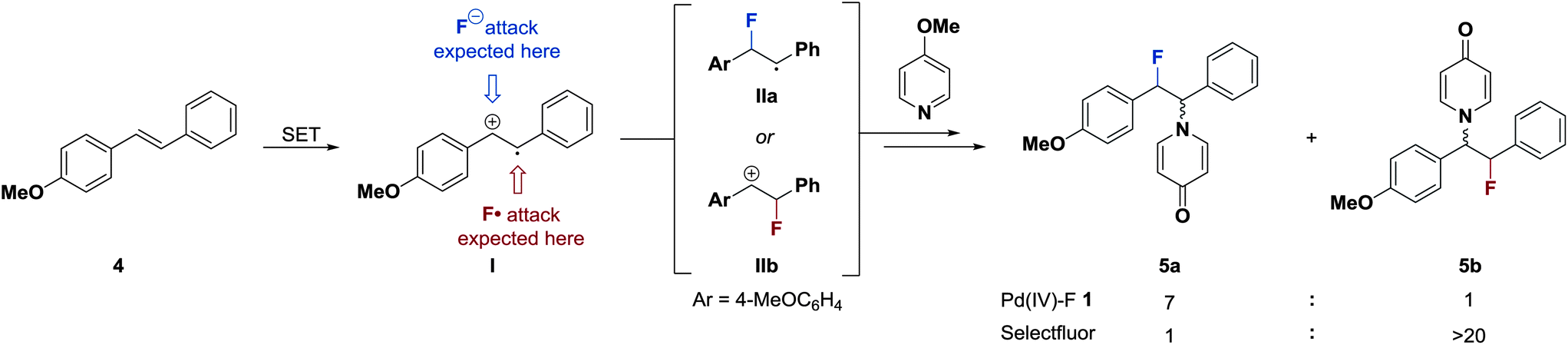
Mechanism of electrophilic fluorination with Pd(iv): fluoride capture and subsequent oxidative fluoride transfer - Chemical Science (RSC Publishing)

Figure 10 from Mechanism of C-F reductive elimination from palladium(IV) fluorides. | Semantic Scholar

Figure 10 from Mechanism of C-F reductive elimination from palladium(IV) fluorides. | Semantic Scholar
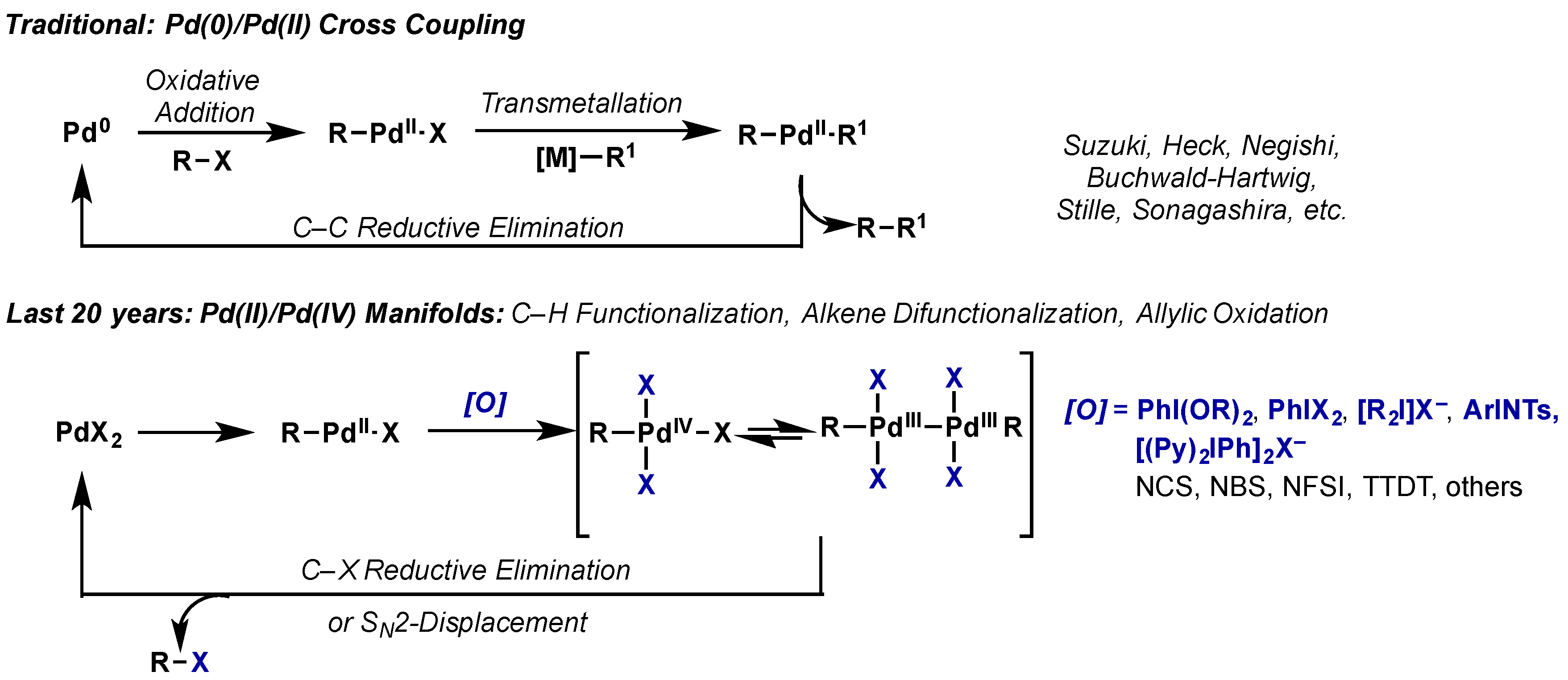
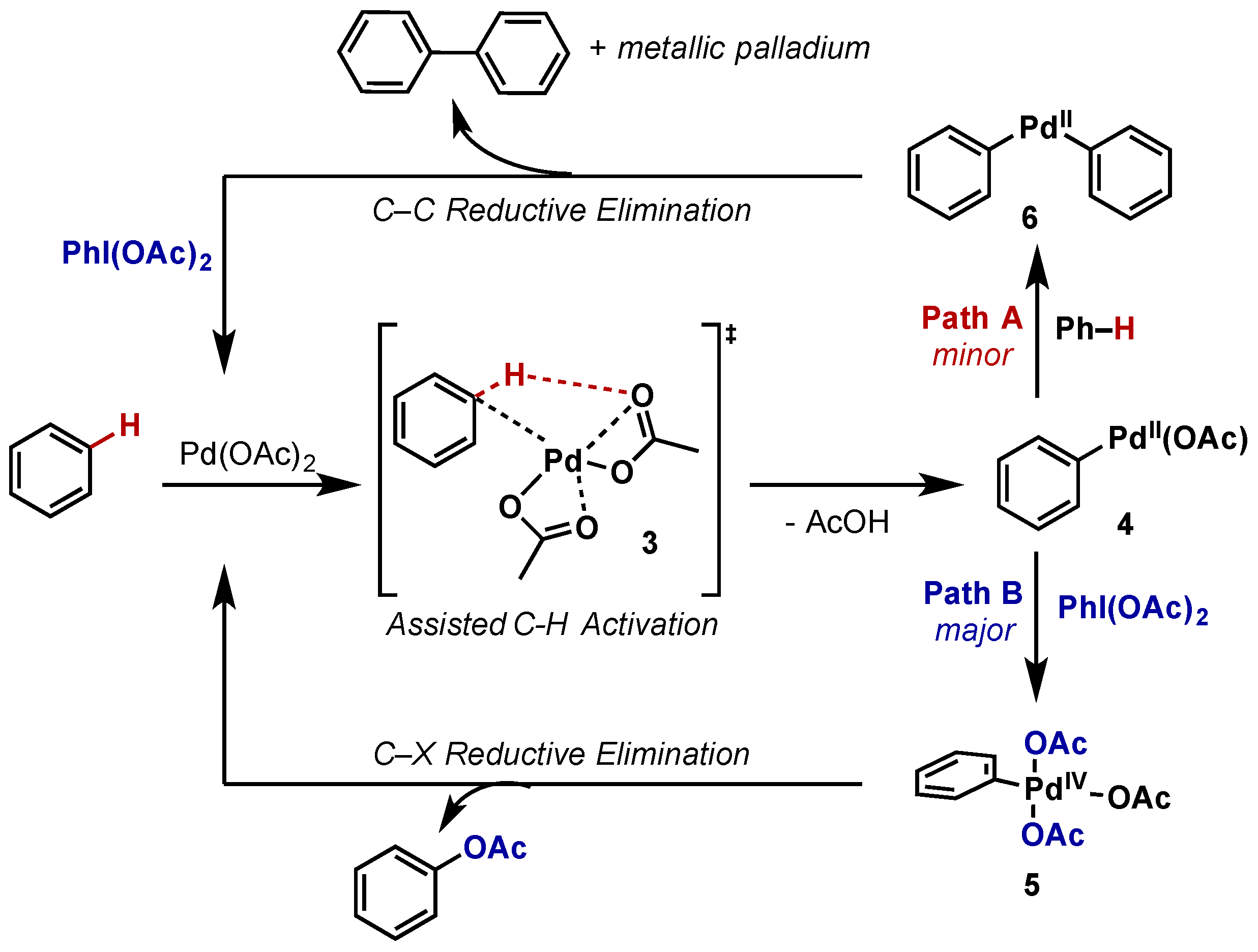
Molecules | Free Full-Text | Hypervalent Iodine Reagents in High Valent Transition Metal Chemistry | HTML

Ligand‐Assisted Palladium(II)/(IV) Oxidation for sp3 CH Fluorination - Sun - 2016 - Advanced Synthesis & Catalysis - Wiley Online Library
Carbon–hydrogen (C–H) bond activation at Pd IV : a Frontier in C–H functionalization catalysis - Chemical Science (RSC Publishing) DOI:10.1039/C4SC02591A

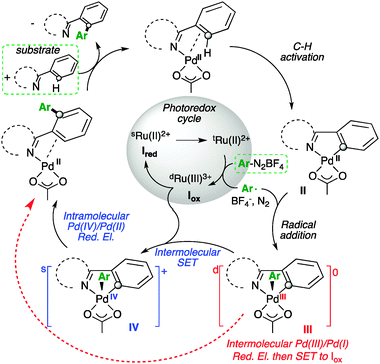
Mechanism of the palladium(II/IV)-catalysed intermolecular C–H amination. | Download Scientific Diagram

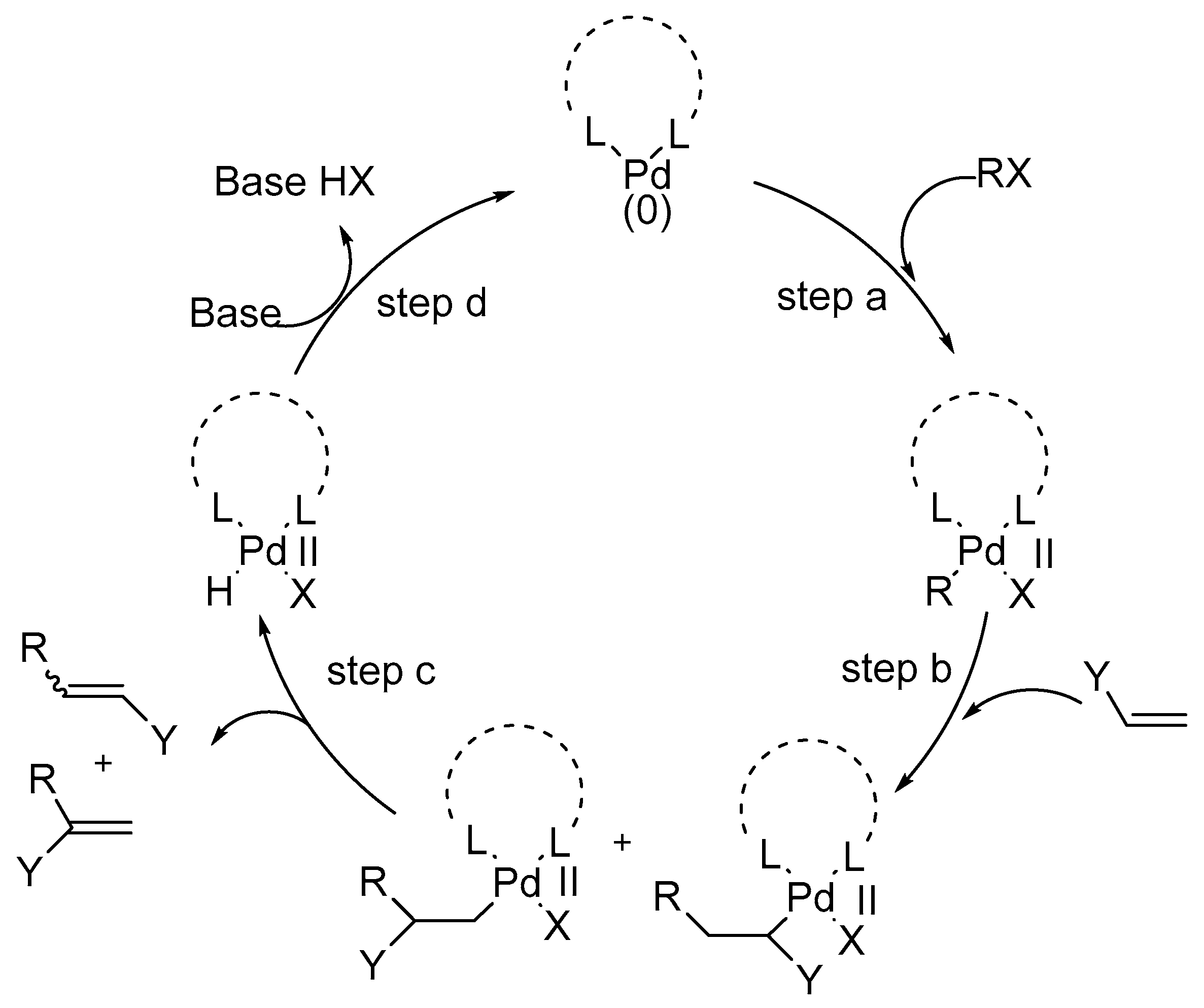
Report: Toward Greater Understanding and Expanded Utility of the Palladium-Catalyzed Activation of Carbon-Carbon Single Bonds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)

Insight into the palladium-catalyzed oxidative arylation of benzofuran: heteropoly acid oxidants evoke a Pd(II)/Pd(IV) mechanism - ScienceDirect

Proposed mechanism for Pd-catalysed oxidative coupling of 2aryl pyridines. | Download Scientific Diagram

Selective Reductive Elimination at Alkyl Palladium(IV) by Dissociative Ligand Ionization: Catalytic C(sp3)−H Amination to Azetidines - Nappi - 2018 - Angewandte Chemie International Edition - Wiley Online Library