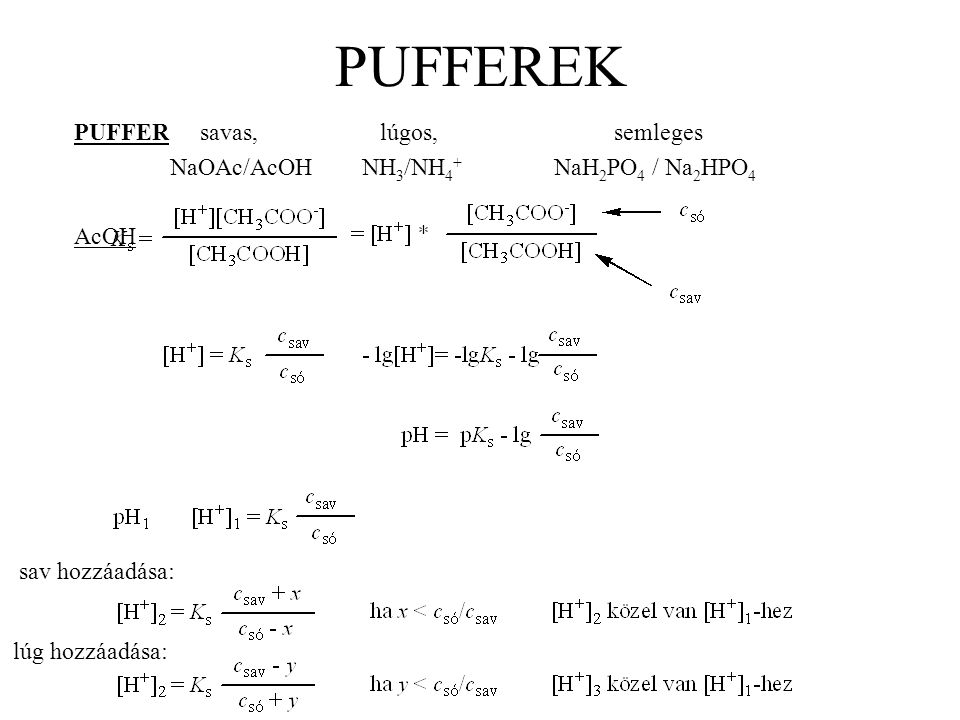
A DNAzyme‐Based Colorimetric Paper Sensor for Helicobacter pylori - Ali - 2019 - Angewandte Chemie - Wiley Online Library

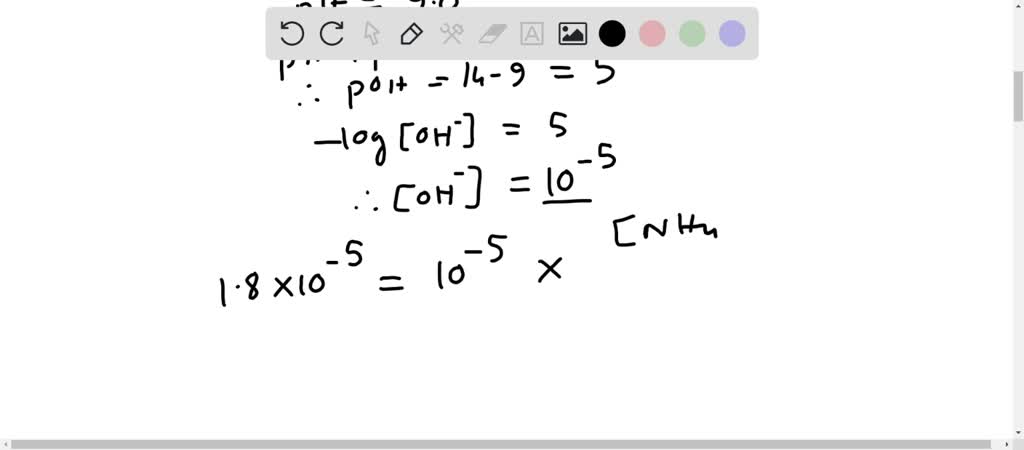
SOLVED: Which solution has the greatest buffering capacity?A) 0.335 M NH3 and 0.100 M NH4ClB) 0.085 M NH3 and 0.090 M NH4ClC) 0.540 M NH, and 0.550 M NH4ClD) 0.200 M NH,

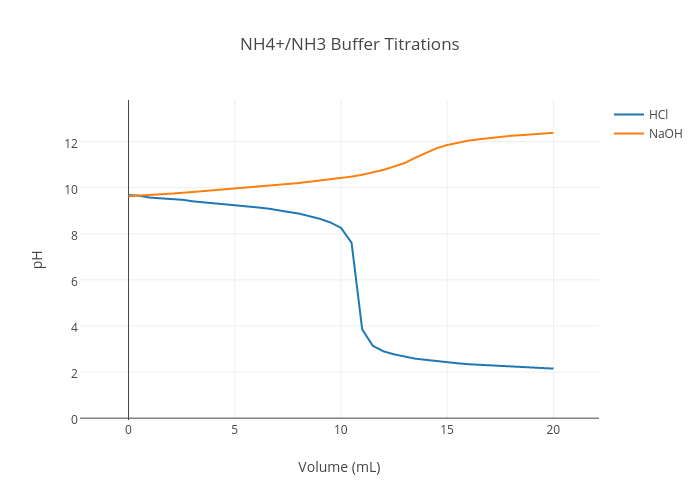
Abb. 2.4: Pufferbereiche der Pufferkapazitäten des VFA-, HCO 3 --und NH... | Download Scientific Diagram
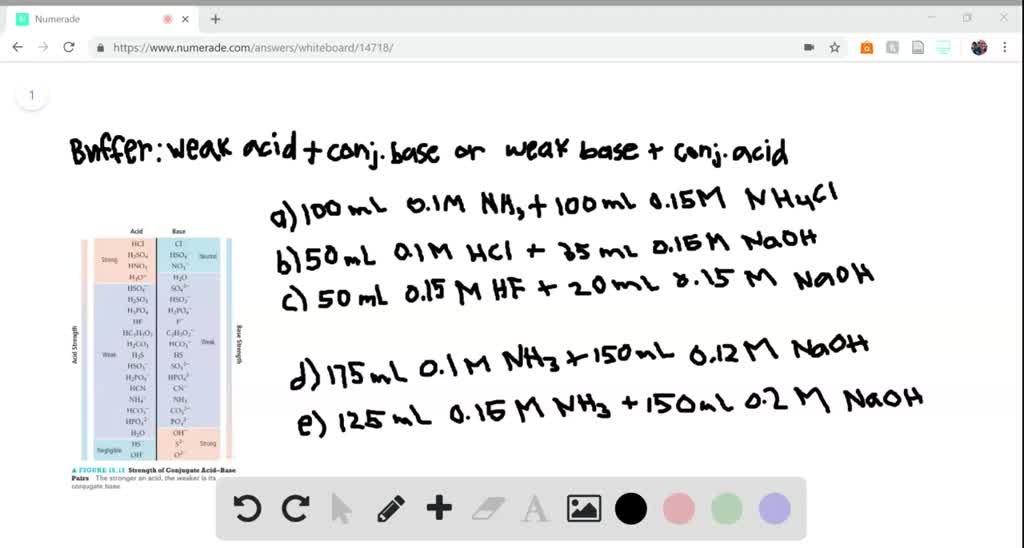
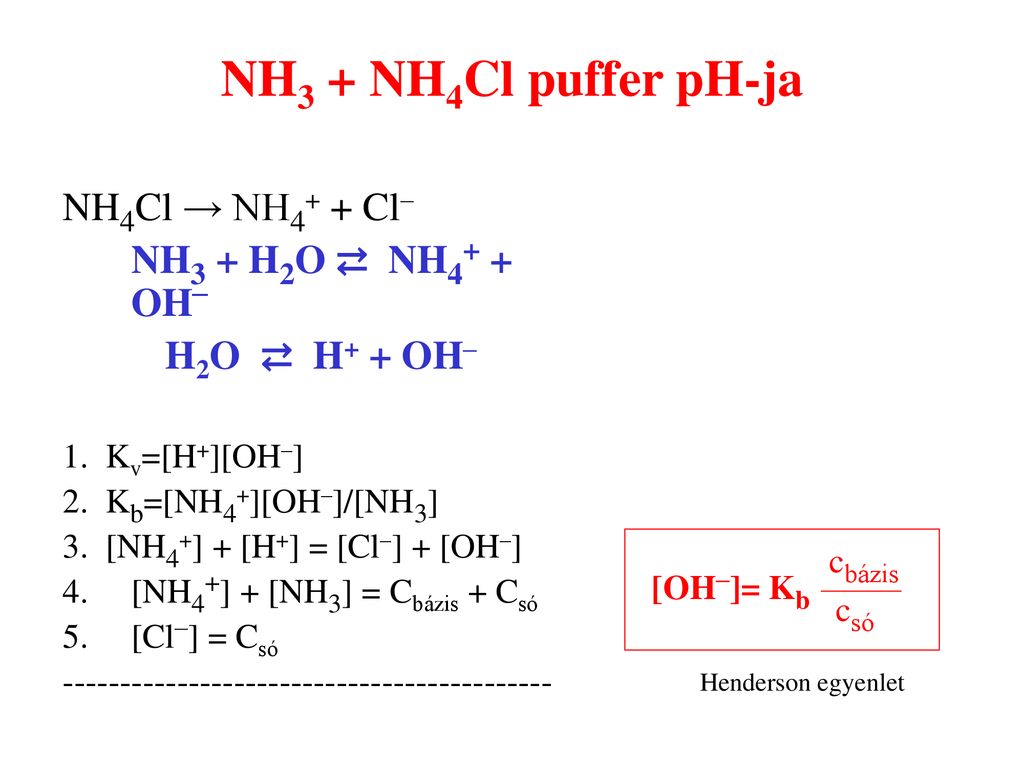
SOLVED:Determine whether the mixing of each pair of solutions results in a buffer. a. 100.0 mL of 0.10 M NH3; 100.0 mL of 0.15 M NH4Cl b. 50.0 mL of 0.10 M

Does anyone have any stocking suggestions? 30 gal, I'm pretty sure I'm all cycled pH 7.2, NH3 0ppm, NO2- 0ppm and NO3 0ppm. : r/Aquariums