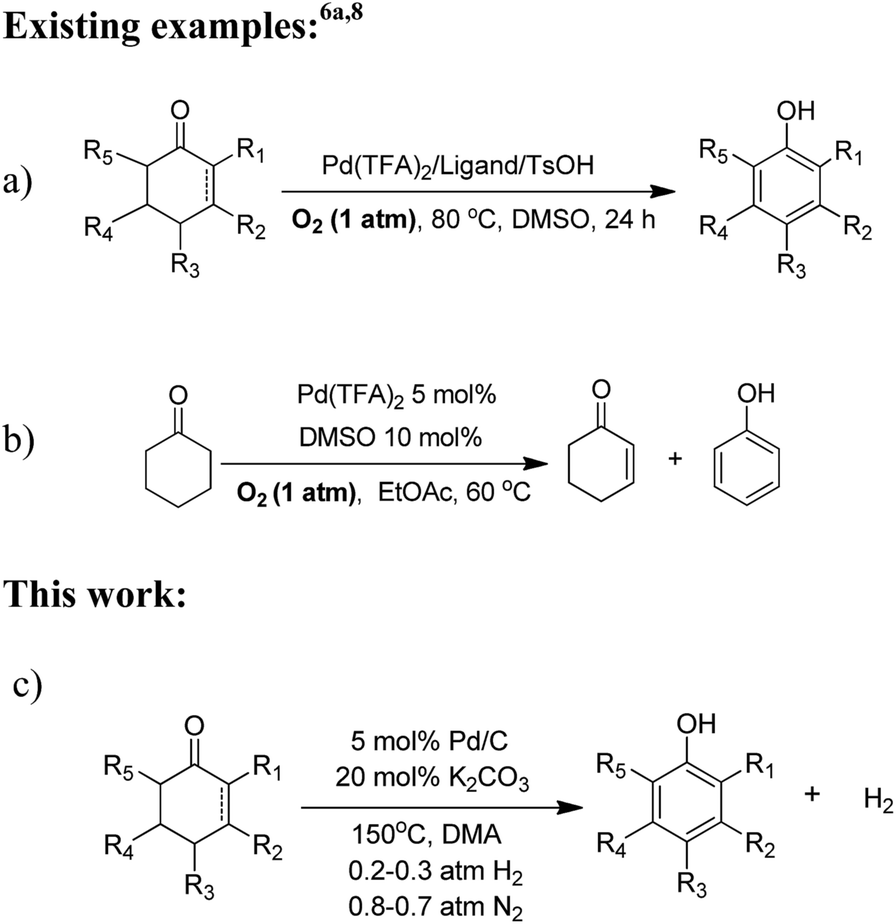
Reaction-activated palladium catalyst for dehydrogenation of substituted cyclohexanones to phenols and H 2 without oxidants and hydrogen acceptors - Chemical Science (RSC Publishing) DOI:10.1039/C5SC01044F

The application of a supported palladium catalyst for the hydrogenation of aromatic nitriles - ScienceDirect

PDF) Pd/P( t -Bu) 3 : A Mild Catalyst for Selective Reduction of Alkenes under Transfer-Hydrogenation Conditions

The application of a supported palladium catalyst for the hydrogenation of aromatic nitriles - ScienceDirect
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img02.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
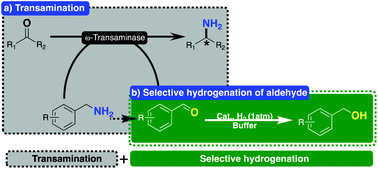
Highly selective hydrogenation of aldehydes promoted by a palladium-based catalyst and its application in equilibrium displacement in a one-enzyme procedure using ω-transaminase - Organic Chemistry Frontiers (RSC Publishing)

Chemoselective control of hydrogenation among aromatic carbonyl and benzyl alcohol derivatives using Pd/C(en) catalyst - ScienceDirect
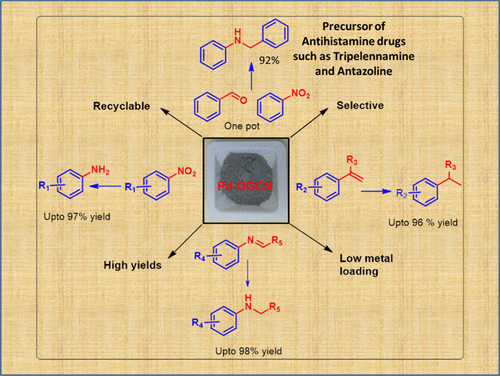
Functionalized Graphitic Carbon Nitride Decorated with Palladium: an Efficient Heterogeneous Catalyst for Hydrogenation Reactions Using KHCO2 as a Mild and Noncorrosive Source of Hydrogen. - ACS Omega - X-MOL

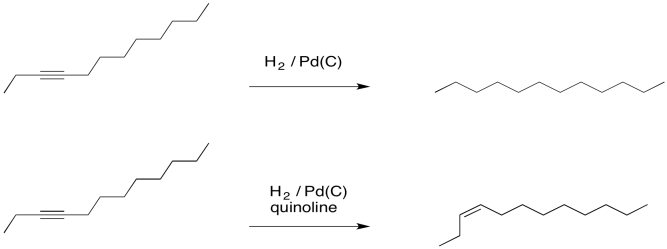
Development of a Palladium‐Catalyzed Process for the Synthesis of Z‐Alkenes by Sequential Sonogashira–Hydrogenation Reaction - Hancker - 2018 - European Journal of Organic Chemistry - Wiley Online Library

Active palladium catalyst preparation for hydrogenation reactions of nitrobenzene, olefin and aldehyde derivatives - ScienceDirect
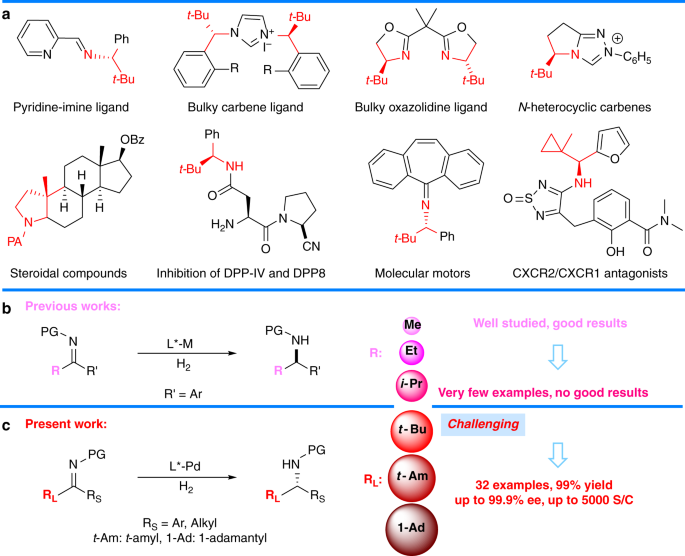



.jpg?revision=1&size=bestfit&width=520&height=939)